Medium Pressure UV vs. Low Pressure UV in Indoor Swimming Pools
Ultraviolet (UV) systems are proven and effective secondary disinfection systems. They are used in a multitude of water treatment applications, like drinking water, wastewater, food processing, and of course, swimming pools.
Covered in this article:
- Two types of UV disinfection systems
- Low pressure UV
- Advantages of low pressure UV
- Disadvantages of low pressure UV
- Medium pressure UV
- Advantages of medium pressure UV
- Disadvantages of medium pressure UV
- Conclusion
Two types of UV disinfection systems
There are two types of UV disinfection systems: low-pressure and medium-pressure.
We know that both types of UV can help water chemistry, yet both types of UV are limited when it comes to improving indoor air quality. UV is a point-of-contact system that creates no residual sanitizer. And to be fair, no secondary disinfection system does...that's why chlorine is still necessary in pools.
Related: Pool Chemistry Resources
We like to think of these two UV systems as having different levels of power. Technically, the term pressure refers to the pressure of gases inside the UV light bulb. It has nothing to do with flow rates or water pressure that moves through the UV chamber. The higher the pressure, the more UV radiation can be released into the water. And indeed, the lower the pressure, the lower the energy consumption.
Both medium-pressure and low-pressure UV systems inactivate pathogens by disrupting the DNA of the cells, preventing their reproduction, and often killing the cell itself.1 Both low and medium pressure UV light can inactivate bacteria, viruses, and protozoan cysts in a single pass.2
Low Pressure UV

Beyond inactivating germs and viruses in a single pass, low pressure UV can destroy monochloramine (NH2Cl) in one pass, through a process called photolysis, or photodegradation.3 If a monochloramine is destroyed, that monochloramine cannot progress and become dichloramine (NHCl2) or trichloramine (NCl3).
Because of this, some low pressure UV manufacturers are proud to say they destroy chloramines, which is partially true, but often misleads people into thinking low-pressure UV can destroy all chloramines.
In reality, no UV system can destroy all monochloramine in the first place, because UV systems are point-of-contact only. Monochloramines out in the pool may have enough time to be oxidized by chlorine and convert into dichloramine and trichloramine without the UV system ever having a chance to see them. And when trichloramines off-gas into the air, they are now an air problem...there's nothing UV can do about it.

Water does not 'wait its turn' to be recirculated and pass through the pump room, filter and UV. Water circulates, gets treated, and mixes back in with the rest of the pool.
So while it is true that monochloramines are destroyed by low pressure UV, it is not true that the pool will have no dichloramine or trichloramine produced. These pools can still have indoor air quality problems. That being said, low pressure UV is proven to reduce combined chlorine levels, at least somewhat.
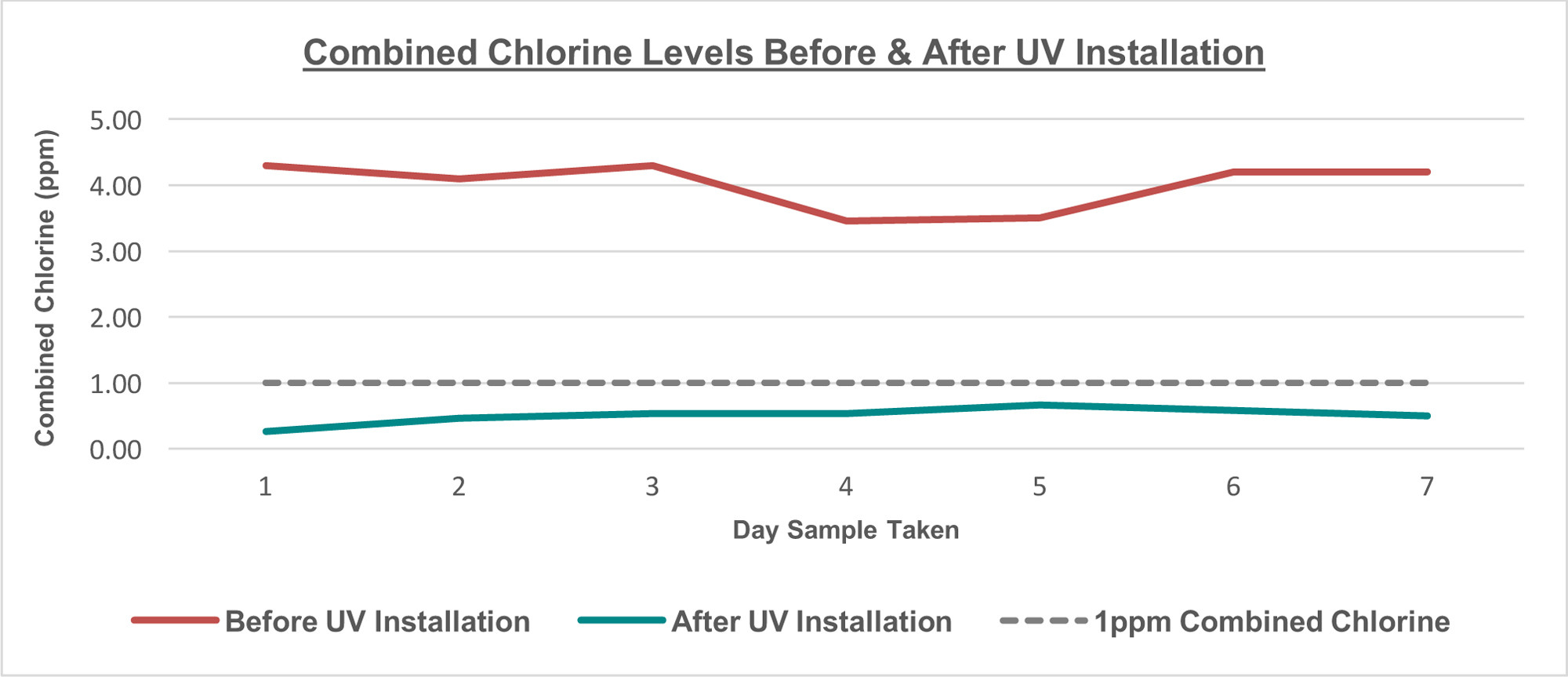
Source: UVGuard.com
Advantages of Low Pressure UV
For the sake of comparison, there are some advantages to low pressure UV. First and foremost is cost. Low pressure UV systems cost significantly less than medium pressure systems, both up front and ongoing. The lower pressure and heat (low pressure UV systems heat up to about 115º-140ºF) means plastic can be used instead of stainless steel. The lamps last longer and use less energy, and therefore require less frequent replacement of bulbs. All of those contribute to a much more affordable system compared to medium pressure UV.
Another advantage that comes with less energy, heat and longer bulb life is that low pressure UV can better handle intermittent flow, meaning they can be used on pools that do not circulate 24/7, or do not circulate at the same rate 24/7. This can be seen as an advantage over medium pressure UV systems, which need constant flow.
The final advantage we will mention is that low pressure UV does not have the wavelength of UV light that destroys free chlorine–or more specifically, Hypochlorous Acid (HOCl). Medium pressure does.

As you can see from that graphic, at 295nm, free chlorine (HOCl) gets destroyed by medium pressure UV, but not low pressure UV. And yet low pressure UV outputs some of the optimal germicidal wavelengths to disinfect water.
Disadvantages of Low Pressure UV
While it can destroy monochloramine, low pressure does not destroy dichloramine or trichloramine. Another disadvantage is against sunscreen, a staple of outdoor pool contamination. Sunscreen blocks UV light by its very nature, so naturally it will interfere with the efficacy of a UV system–especially if it builds up on the glass sleeve around the bulb itself. UV can only inactivate what its light actually penetrates, and if sunscreen is blocking that light, less water will be fully exposed to the UV rays.
Medium Pressure UV
 Medium Pressure UV systems create more heat and pressure, and are therefore must be made of stainless steel, not plastic.
Medium Pressure UV systems create more heat and pressure, and are therefore must be made of stainless steel, not plastic.
While low pressure UV is more popular on residential pools, medium pressure UV dominates in the commercial pool market. Without a doubt, medium pressure UV is the most widely-used secondary system for indoor swimming pools. It's not even close.
Advantages of Medium Pressure UV
The main advancement that medium pressure offers is the amount of energy that a single system can put into the water. This energy difference is evident by the sheer size of the power supply box and the fact that medium pressure UV systems are made of thick stainless steel, not plastic. They put out enough heat to eventually melt PVC. Fortunately, water is moving quickly through the chamber, so it is constantly being cooled by flowing water.
Medium pressure has much more power, and therefore can inactivate more things in the same amount of water, faster. This allows the system to have a smaller physical footprint with less lamps, whereas a low pressure system may need multiple lamps to equal the same rate of disinfection of medium pressure.
But the main advantage of medium pressure UV is its broader spectrum of UV light wavelengths. This means medium pressure systems can inactivate and destroy more contaminants. Medium pressure UV has been proven to inactivate cryposporidium, giardia, and other dangerous recreational water illnesses (RWIs) with ease. 200-230 nm wavelengths, called Far UV, is effective at inactivating viruses like COVID-19 (specifically centered around 222 nm).4
And we're also talking about things beyond common germs. As mentioned before, low pressure UV can only destroy monochloramine (NH2Cl). Medium pressure UV, on the other hand, destroys all inorganic chloramines in the water, and potentially some other disinfection byproducts (DBPs) too.
Disadvantages of Medium Pressure UV
Medium pressure systems require constant flow, as the bulbs release a lot of heat (medium pressure UV systems heat up to 1100ºF). This usually means the bulbs have a shorter useful lifespan. Cooling is necessary, and if water is not constantly flowing, it can shorten the lifespan of the bulbs even more. This contributes to higher operating costs, which we'll expand upon in a moment.
If you look at the wavelength diagram above, you will see that medium pressure UV does not only destroy chloramines. It also destroys free chlorine (HOCl). According to an article in Wastewater Digest:
High intensity, broad-spectrum UV systems (also known as medium-pressure UV) reduce both free chlorine and combined chlorine compounds (chloramines) into easily removed byproducts.
Between the wavelengths 180 and 400 nm, UV light produces photochemical reactions that dissociate free chlorine to form hydrochloric acid. The peak wavelengths for dissociation of free chlorine range from 180 to 200 nm, while the peak wavelengths for dissociation of chloramines (mono-, di- and tri-chloramine) range from 245 to 365 nm.5
And while destroying free chlorine and its byproducts can be beneficial for drinking water, we need free chlorine in our swimming pools as a primary sanitizer. To us, that seems like it's a conflicting strategy. Direct sunlight, of course, also destroys free chlorine. This is why cyanuric acid (CYA) is used in outdoor pools, but not indoor pools.
But chlorine can be replenished, and most commercial pools that have medium pressure UV have chemical automation and chlorine feeders.
Perhaps the most important disadvantage of medium pressure UV compared to low pressure UV is its cost. Expensive to purchase, expensive to install, expensive to operate (energy), expensive to maintain (bulb cleaning and replacement). But then again, when comparing to other secondary disinfection systems like Ozone and AOP, it's in the same order of magnitude. Great water quality is not cheap.
Another disadvantage is the fact that medium pressure UV systems are made of metal, which is susceptible to corrosion in the pump room. This can be very costly. The corrosion issue in pump rooms is one that we take seriously, because ventilation is critically important in pump and chemical storage rooms too.
Related: Got corrosion in your pool or pump room?

And finally, just like all secondary systems, medium pressure UV cannot treat what it cannot reach. Therefore it has limited benefit to indoor air quality. Yes, it helps, but it's not enough. Most of our clients with indoor air quality problems have had medium pressure UV for years, and they still struggle with chloramines in the air.
Conclusion
Both low and medium pressure UV systems improve water quality, and have limited impact on indoor air quality. They are excellent secondary disinfection systems and they leave behind virtually no byproducts. From our standpoint, UV disinfection is strongly recommended for indoor pools.
UV systems are effective germicides, especially at their optimal disinfection wavelength of 264 nm. Both low pressure and medium pressure systems can deliver that wavelength.
1 Sholtes, K., Lowe, K., Walters, G., et.al. (2016). Comparison of ultraviolet light-emitting diodes and low-pressure mercury-arc lamps for disinfection of water. Environmental Technology, 37 (17), 2183-2188.
2 Linden, K., Sobsey, M. (2005). Final Report: Effectiveness of UV irradiation for pathogen inactivation in surface waters. US Environmental Protection Agency. EPA Grant #R829012.
3 Watts, M., Linden, K. (2007). Chlorine photolysis and subsequent OH radical production during UV treatment of chlorinated water. Water Research, 41 (13), 2871-2878.
4 Blatchley, E., Brenner, D., Linden, K., et.al. (2022). Far UV-C radiation: An emerging tool for pandemic control. Critical Reviews in Environmental Science and Technology, 53 (6), 733-753.
5 McClean, J. (2021). Using UV for dechlorination. Wastewater Digest.

 By
By


