Could You be Allergic to Chlorine?
Is it possible to be allergic to chlorine? No. But swimmers, coaches and lifeguards who spend a lot of time in natatoriums seem to think so. Believe it or not, your body produces its own chlorine (specifically Hypochlorous acid, or HOCl) as part of your immune system.
So if it's not a chlorine allergy, what's actually going on? Why is swimming indoors so irritating, itchy, burning and uncomfortable? Why do so many swimmers and lifeguards cough? Swimmers mistakenly blame the irritation they feel on chlorine, when it's actually the byproducts of chlorine, called chloramines.
Covered in this article:
- Personal experience: diagnosed with a chlorine allergy
-
Understanding the human immune system
- How does the human body create its own chlorine?
- What is an allergy?
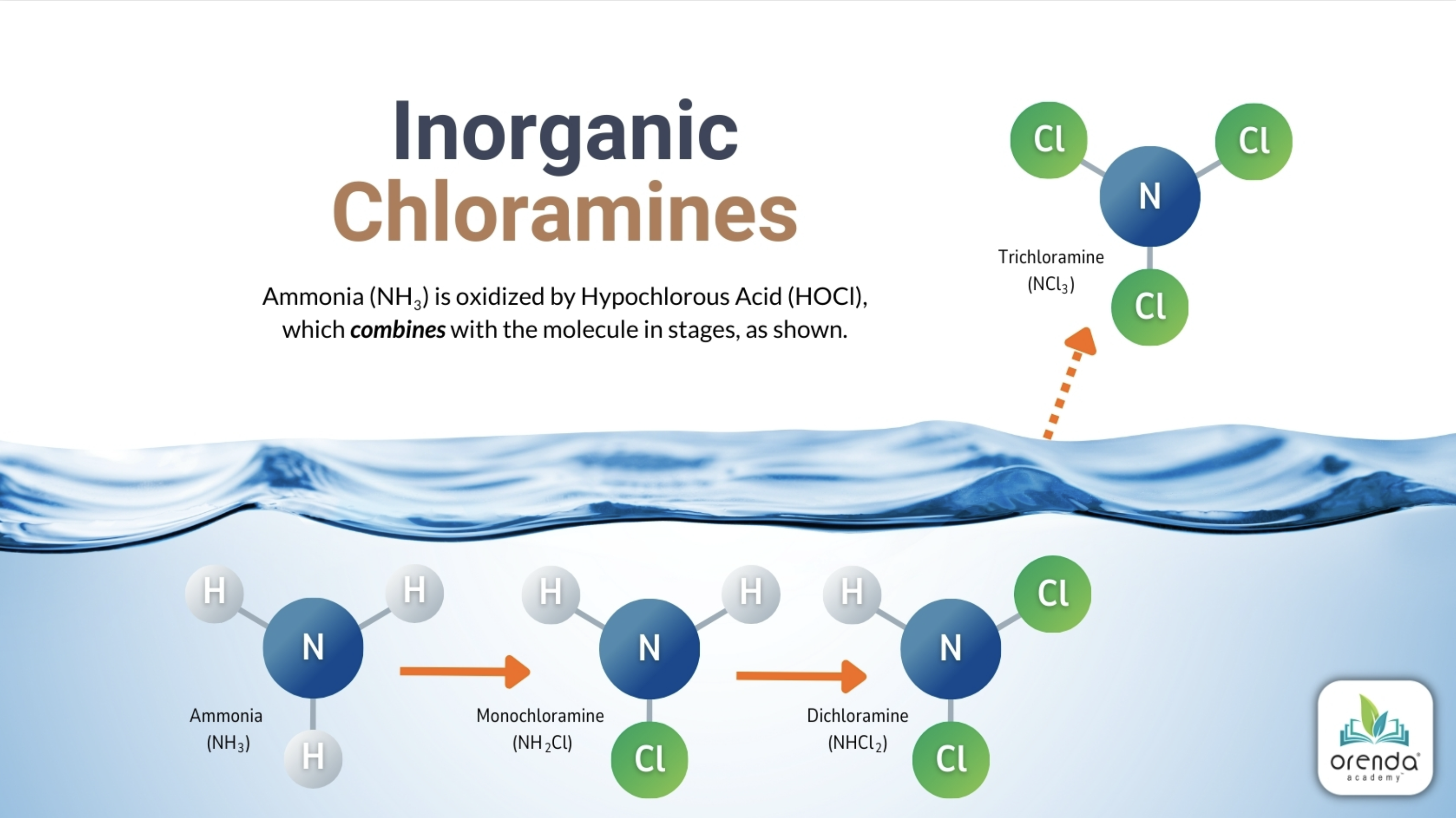
- Chloramines: The Real Source of Swimmer Discomfort
- Our Approach: How to Reduce "Chlorine Allergy" Symptoms in Your Natatorium
- Conclusion
I had the privilege of speaking with Dr. Michael Fields, a pediatric pulmonologist who reached out to us based on this website. He contacted us because he sees many swimmers who struggle with breathing in indoor pools due to bad air quality. You can listen to our podcast episode above.
Dr. Fields inspired me to do more medical research into this. And given that I am not a doctor, I have no intention of diagnosing any issues you may have. Instead, I want to lay out what the research and science says, and let you talk to your own doctor about it.
Personal Experience: Diagnosed with a Chlorine Allergy
But first, my story. I swam in college, and at the time, it was a bromine pool. I had no breathing or skin irritation issues (though I will never forget that distinct bromine smell). During the summer after my sophomore year, the university recreation department decided to switch the sanitizer to calcium hypochlorite, which required a complete drain and refill of the 1.2 million gallon olympic sized pool we had.
When we returned to training in late August, breathing issues arose. I was in a constant state of mild to moderate sickness. The lymph nodes in my neck were swollen more often than not, and I found myself coughing while swimming, and sometimes randomly throughout the day, like having coughing fits during classes. Things reached a breaking point for me when I was unable to complete threshold sets because my lungs were burning so bad I wasn't able to breathe fully.
So I went to the athletic training office, and once a week they'd have a physician there for student athletes. I made my appointment, and the doctor told me I had all the symptoms of sport-induced asthma, or exercise induced bronchoconstriction (EIB).1
That same doctor also told me I must be allergic to chlorine, because there was also burning eyes, itchy skin and other allergy symptoms.
At the time, I was on a partial scholarship for swimming. Quitting the sport was simply not an option if I wanted to stay in school. My next best option was a prescription for an albuterol inhaler.
For years, I believed I was allergic to chlorine, and it lingered in the back of my mind that my timeline in the sport was going to be short-lived. I did not want to compromise my health long term.
But then again, every time I swam outdoors, I had zero issues. No itchy skin, watery eyes, burning lungs. No breathing issues. I could train hard and make it through the workout. In hindsight, I should have picked up on what was going on, but at the time, I was so focused on stroke technique and my training that I wasn't paying attention to these things.
Fast forward to today. I am writing this article because I was one of many who believed I was allergic to chlorine. And I'm happy to tell you, such an allergy does not exist. Irritation from chloramines and other disinfection byproducts, however, is very real. And that's what I was dealing with.
Understanding the Human Immune System
I have researched chloramine-related health issues for years. Most of it is about how chloramines do us harm, and that much is well documented. I was surprised, however, to learn that our own immune systems produce chlorine. Specifically, a type of white blood cells–called neutrophils–utilize an enzyme called myeloperoxidase.2
While white blood cells have a variety of ways of killing pathogens, producing chlorine is one of them.
How does the human body create its own chlorine?
When neutrophils meet a pathogen (a germ or virus, for instance), they combine chloride ions and hydrogen peroxide in our blood to create chlorine in the form of Hypochlorous Acid (HOCl).3 This "oxidative burst" destroys the germs.
But where do the chlorides come from in the first place?
We have chloride ions in our blood because we consume salt with our food (sodium chloride, NaCl). And about 80% of our bodies are made of water. When I teach this concept in the Watershape University chemistry class (Service 2211: Essential Water Chemistry), I relate this process to how a salt chlorine generator works.4
Indeed, we humans are biochemically engineered to produce our own chlorine, therefore it is considered endogenous.5 And because it's endogenous, our bodies have evolved to manage chlorine through various antioxidant pathways and metabolic processes so it doesn't harm our own tissue. All that is to say, this is why humans cannot be allergic to chlorine.6
What is an allergy?
Technically, in the field of immunology, an allergy is defined as an Exaggerated immunoglobulin E (IgE)-mediated hypersensitivity reaction:
"Type 1 or immediate hypersensitivity is an exaggerated immunoglobulin E (IgE)-mediated immune response to allergens such as pollen, dust mites, foods, medications or insect venom, affecting nearly one-third off the global population. The reaction occurs when antigen-specific IgE binds to mast cells and basophils, triggering the release of inflammatory mediators, including histamine, tryptase, and proteases. Reactions may be immediate, delayed, or the cause of chronic allergic inflammation, as observed in sensitized individuals with ongoing exposure, such as pet owners with animal allergies. Clinical manifestations range from mild symptoms, including allergic rhinitis, urticaria, and atopic dermatitis, to severe, potentially life-threateninganaphylaxis requiring immediate care." 7
If you cannot be allergic to chlorine itself, can you be allergic to chloramines, since they are known irritants?8 The answer is yes. And worse yet, because inhaling chloramine vapor irritates the lining of our lungs (Called the pulmonary epithelium), it makes our lungs more susceptible to other allergens and irritants.
Chloramines: The Real Source of Swimmer Discomfort
Field experience and extensive research both make it clear that disinfection byproducts like chloramines are the root cause of most swimmer discomfort. Sure, excessive chlorine levels can irritate swimmers, but that's not an allergic reaction.
Several other things are mistakenly blamed for irritation and discomfort, including an unbalanced pH.
Despite what many legacy sources have said, we have not seen any evidence that swimming in water with a pH between 5.5 and 9.5 has any impact on bather comfort or eye irritation. The belief is that the pH of our eyes is 7.4, so if the water is too far from that, it will cause irritation. But according to a study published by the National Institutes of Health (NIH), the pH of our tears can vary based on how hydrated we are in that moment.9
I personally have had the opportunity to race in swim meets in Europe, where the pools maintain a 6.5 pH and a relatively low Total Alkalinity, but offset it with high calcium hardness. These pools follow the European DIN standard, which means much less free chlorine than we are accustomed to in the United States. Guess what? I had no itchy skin or irritated eyes.
Indeed, it is chloramines and other harmful byproducts that are the irritants.10
Our Approach: How to Reduce "Chlorine Allergy" Symptoms in Your Natatorium
As you may know by now (if you've been reading our website), disinfection byproducts are inevitable when swimmers use the pool. The question is what can you do about them? We believe in a four-pronged approach:
-
Identify and limit the sources of nitrogen before they get in the pool,
-
Address non-living organics in the water by using enzymes,
-
Supplement chlorine with a secondary system like ozone or UV, and
-
Optimize airflow design in the natatorium (which is our specialty here).
Let's expand on each of them briefly.
1. Identify and limit the sources of nitrogen before they get into the pool
High combined chlorine levels (above 0.5 ppm) are rare in pools that only have human sources of nitrogen (like sweat and urine). There is usually a nitrogen-based chemical getting into the water somehow. Normally, it's either an ammonia-based deck cleaner, or algaecide.
Remove any such chemicals from the facility, even if they're not used in the pool area specifically. Cleaning the locker room with these chemicals will inevitably get it tracked back into the pool by wet swimmers.
2. Address non-living organics in the water by using enzymes
Chlorine is an incredible disinfectant, but a comparatively lousy oxidizer. Chlorine was made to kill germs, not to oxidize bather waste. Non-living organics have carbon bonds, and at the chlorine levels used in pools, chlorine is simply not strong enough to break down those bonds. The consequence? Partially-oxidized contaminant byproducts; many of which are harmful and cause irritation.
Enzymes do NOT break down nitrogen, but if it's organic nitrogen, enzymes can take the carbon out of the equation, breaking down the complex molecule into simpler components. This greatly reduces the energy levels required for chlorine to destroy the contaminant through the combined chlorine process.
Below shows the comparison of inorganic nitrogen on the left, versus organic nitrogen on the right. As you can see, urea is much more complex.

Enzymes can break urea into inorganic ammonia, which chlorine would then have to deal with. Chlorine manages inorganic ammonia by combining with it to form chloramines. Hence the name combined chlorine.

So help chlorine out! Get carbon out of the water with enzymes so chlorine can focus on what it needs to to keep your water safe, clean and clear. Besides, removing non-living organics (like oils) does amazing things for water clarity too, that chlorine and filtration alone cannot:

And enzymes are easy to install on a feed pump. Pound for pound, they are one of the most easily-implemented and affordable solutions to improving air quality.

3. Supplement Chlorine with a Secondary System like Ozone or UV
Expanding on previous point, enzymes can only address carbon. A secondary oxidizer like ozone or AOP can destroy just about anything. And Ultraviolet (UV) systems can add a layer of secondary disinfection too (though they do not oxidize).
Low-pressure UV systems can break the chlorine-to-ammonia bond, to break apart monochloramine. Medium-pressure systems are more robust, expensive, and create much more heat. they also create a much wider spectrum of light, and can also break apart dichloramines and trichloramines.

That being said UV systems do not destroy chloramines, they only break the bonds apart. The ammonia can combine with more chlorine in the future.
Secondary systems in general are great, but they are all point-of-contact systems, and do not put a viable residual into the water. That's why chlorine is the primary sanitizer on almost all commercial pools in the United States.
INSIGHTS FROM OUR EXPERIENCE:
IN THE 360+ NATATORIUMS WE HAVE EVALUATED, we estimate over 80% of our clients already had a secondary disinfection system like UV, ozone or AOP. And yet, they hired us because they still had bad air quality.
- Of the remaining ~20%, most of them had a UV, Ozone or AOP system, but it was not working at the time of our visit. Neglecting maintenance is a common issue amongst pool operators, unfortunately.
- Less than 10 facilities (out of 360+ that we've visited) had even tried using enzymes.
4. Optimize Airflow Design in the Natatorium
This is what we can help you with. Almost every natatorium we have visited could have had great air, but certain fundamentals were incorrect. In most cases, if we don't correct the airflow, good air quality is impossible, not just difficult.
If you are designing a new natatorium, we can help you avoid these mistakes before the project breaks ground. Our design assistance saves the design engineer a lot of time in the process.
If you have an existing indoor pool that is struggling with bad air or water quality, we offer site visit evaluations, write a detailed report, and work with a local engineer and contractor of your choice to fix the real problems. Not the symptoms; the actual root problems.
Conclusion
No, you're not allergic to chlorine. You produce it in your blood. It's most likely disinfection byproducts like chloramines that are causing the symptoms. And while they are inevitable in some ways, our four-pronged approach has proven to mitigate the issues substantially.
If you are struggling with these issues, perhaps we can help your facility too. Because chloramine problems won't solve themselves.
1 Boraczyński, M., Balcerek, T., Rożkiewicz, N., Pabiszczak, M., Harasymczuk, M., Sławska, A., & Lutomski, P. (2025). Pulmonary function in swimmers exposed to disinfection by-products: a narrative review. Frontiers in Physiology, 15. https://doi.org/10.3389/fphys.2024.1473302
2 Rosen, H., Crowley, J. R., & Heinecke, J. W. (2002). Human neutrophils use the myeloperoxidase-hydrogen peroxide-chloride system to chlorinate but not nitrate bacterial proteins during phagocytosis. The Journal of biological chemistry, 277(34), 30463–30468. https://doi.org/10.1074/jbc.M202331200
3 Pattison, D. I., Hawkins, C. L., & Davies, M. J. (2007). Hypochlorous acid-mediated protein oxidation: how important are chloramine transfer reactions and protein tertiary structure?. Biochemistry, 46(34), 9853–9864. https://doi.org/10.1021/bi7008294
4 A salt chlorine generator uses electricity to create chlorine through a process called electrolysis, whereas our bodies utilize enzymes to make the conversion.
5 Endogenous is a fancy way of describing something that originates internally.
6 Chlorine is the most commonly used disinfectant in the world (by far), and is used in almost all drinking water treatment processes in the developed world. Chlorine itself is not the problem, in the doses we use it in. Obviously high doses can be harmful, but that's not related to whether or not humans can have a chlorine allergy.
7 Abbas M, Goldin J. (2025). Type I Hypersensitivity Reaction. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK560561/
8 American College of Allergy, Asthma & Immunology. (n.d.). Chlorine Allergy | Causes, Symptoms & Treatment. Retrieved from https://acaai.org/allergies/allergic-conditions/chlorine-allergy/
9 Abelson, M. B., Udell, I. J., & Weston, J. H. (1981). Normal human tear pH by direct measurement. Archives of ophthalmology (Chicago, Ill. : 1960), 99(2), 301. https://doi.org/10.1001/archopht.1981.03930010303017
10 Jacobs, J. H., Spaan, S., van Rooy, G. B., Meliefste, C., Zaat, V. A., Rooyackers, J. M., & Heederik, D. (2007). Exposure to trichloramine and respiratory symptoms in indoor swimming pool workers. European Respiratory Journal, 29(4), 690–698. https://publications.ersnet.org/content/erj/29/4/690

 By
By