Pool Deck Cleaners and other Ammonia Sources
Got combined chlorine in your indoor swimming pool? If so, you're not alone. And not all combined chlorine issues can be blamed on swimmers peeing in the pool or not showering. Oftentimes, the type of deck cleaner used can be a source of combined chlorine too. Let's discuss all the ammonia sources that we know of.
Covered in this article:
- How does ammonia get into a swimming pool?
- Chloramines in source water
- Urea from bathers
- Natural sources of nitrogen
- Ammonia-based cleaning products
- Competition pool decks
- Indoor water park (case study)
- Conclusion
How does ammonia get into a swimming pool?
Combined chlorine can be solved if you can figure out one question: how did nitrogen/ammonia get in the pool? There are four common ways ammonia gets into a swimming pool:
Chloramines in source water
Nitrogen compounds like ammonia are sometimes added to drinking water to create chloramines. Municipal water treatment plants sometimes intentionally chloraminate water because combined chlorine lasts longer and has some disinfection power. By adding ammonia to drinking water, disinfection can extend the distance of the water grid. To quote the late pool chemistry expert Robert Lowry:
"Chlorine is used in drinking water treatment. Water treatment plants add ammonia to chlorinated water to make chloramines on purpose. Combining chlorine and ammonia under exact ratios turns all of the free chlorine in the water into combined chlorine. This is mostly monochloramine which does have a foul smell but it also kills. The chlorine-ammonia bond is strong and persists in the water. Chloramines kill organisms but not as quickly as Free Available Chlorine (FAC). However, in drinking water distribution there is usually a long time between chloramination at the municipal water treating facility and the time you use it. Chloramine takes longer than FAC to kill or oxidize, but it does work." 1
Water treatment facilities often add ammonia to drinking water intentionally, because it extends the range of disinfection from treatment to use. There are myriad studies about the impact of chloramines in drinking water and not many health advocates like it. Here are some sources you can check out if you're curious about drinking water chloramines. We won't dwell on it because we are focused in this article about those chloramines and their impact on a swimming pool.
- Chloramines in Drinking Water (EPA)
- Disinfection with Chloramines in Drinking Water (CDC)
- Facts about Chloramine Drinking Water Treatment (Water & Health)
- 5 Red Flags for Chloramination
To remove chloramines from your tap water (if you have them), consider a carbon filter on your fill line to remove chloramines before they get into your swimming pool.
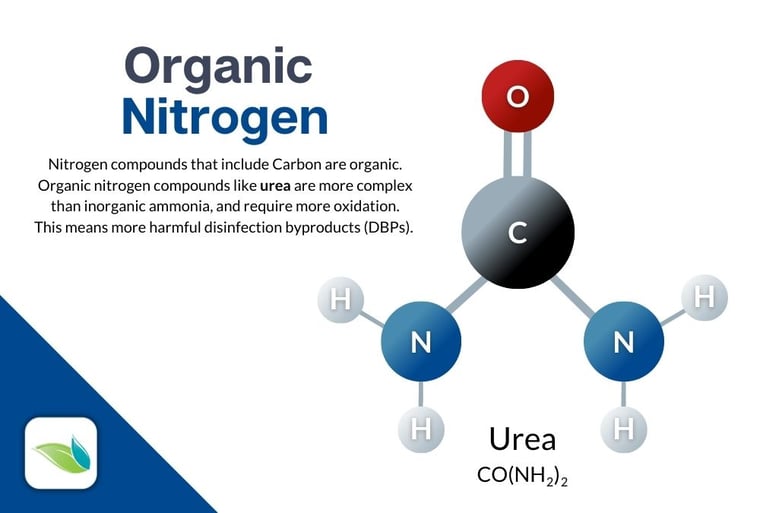
Urea from bathers
People pee in the pool at fairly alarming rates. Check out this video by Mark Rober on YouTube:
But all that being said, it takes a lot of urine to create combined chlorine levels that exceed 0.5 ppm. Modern commercial pools often have secondary disinfection systems like UV, Ozone, or AOP. These systems help chlorine address the urea that comes out of our bodies (urine and sweat). It takes a lot of bathers peeing throughout the day to overwhelm a good swimming pool system.
And hopefully your pool has a lot of swimmers! It's good for business to have a busy pool. A couple of things can chemically help manage bather demand.
First and foremost is to assist chlorine by supplementing it with enzymes to handle non-living organics. Organic bather waste is carbon-based (not nitrogen-based), and makes up an overwhelming majority of the contamination in a typical swimming pool. It makes up a huge majority of the oxidant demand too, followed by not-so-closely by nitrogen compounds and metals. Every pool is different, so exact percentages are undefined.
Enzymes free up chlorine to focus on what it does best: sanitize. Having more free available chlorine gives your pool a better chance of maintaining clean, safe water with low combined-chlorine levels. Why? Because chlorine can more rapidly complete the breakpoint chlorination process to destroy nitrogen compounds and combined chlorine.
Enzymes cannot do anything to nitrogen compounds, unfortunately. Chlorine must oxidize them, or a secondary oxidizer like ozone can.
Natural sources of nitrogen
Sometimes nitrogen compounds get into the pool naturally, such as muddy shoes on the pool deck, which could have been from nitrogen-rich soils. This is more of an issue with outdoor swimming pools than it is with indoor natatoriums, but should still be considered in the realm of possibility. In Europe, outside shoes are required to be taken off, or covered with the same kind of disposable 'booties' that are worn in hospitals. This may seem ridiculous to us in North America, but keeping outside contamination off the pool deck really does make a difference for air quality.
Arguably the best indoor air quality in America is at a small NCAA Division 2 private college called Queens University in Charlotte, NC. They not only have source-capture exhaust and an HVAC design specifically made for optimal air quality; Queens enforces no outdoor shoes on the pool deck. Period. They do many other things too, but it is one of the only pools in America that we have ever been required to remove our shoes before walking on the deck. Perhaps there's something to the idea that nitrogen can come from the bottoms of our shoes. And that leads us to our final source of ammonia: pool deck cleaners.
Ammonia-based cleaning products

Finally, deck cleaning products that contain ammonia are often overlooked. We have seen numerous facilities that used a disinfectant deck cleaner with active ingredients like dimethyl ammonium chloride. Combined chlorine levels were through the roof. But once the cleaning product was swapped out for non-ammonia deck cleaner, the combined chlorine issue vanished within a week.
Competition pool deck
We first noticed this a few years ago at a new, major competition venue that shall remain anonymous. This facility was state-of-the-art in just about every way: 50 meter Olympic sized pool, regenerative DE filters, UV secondary disinfection, the works. Within the first few days of opening, they hosted a swim meet which we attended and competed in. The air quality was awful. Abysmal. Intolerable. And yet, this facility had not even been open for a week. This was the first swim meet!
We later found out that the pool deck is a major source of air quality problems, because chlorinated water splashes and soaks the pool deck. All of us on deck–whether it was coaches wearing shoes or swimmers that were barefoot–were contributing to the organic demand and/or nitrogen compounds that wound up on that pool deck. Chlorinated water oxidizing our waste on that deck led to chloramines. Or so we thought.
It would take a lot of nitrogen from sweaty feet or soils on shoes to get the bad air quality we experienced. Months later, through process of elimination, the facility was asked what deck cleaner product they were using. Sure enough, it was ammonia-based.
So the deck was being scrubbed and disinfected every night with ammonia. The next day, of course, chlorinated water soaked the deck again and reacted with the ammonia to give off unbearable levels of chloramines in the air. Once the deck cleaner was swapped out for a non-ammonia disinfectant product, the air quality became noticeably better almost immediately.
Indoor waterpark with multiple pools
Years went by before we encountered the issue again...or at least until it was brought to our attention again. It was at an indoor waterpark in California that too shall remain anonymous. We were called in because the air quality had some problems, but the real issue was four out of the seven bodies of water had extremely high levels of combined chlorine (> 2.0 ppm, when the health code maximum is 0.2 ppm), ten times legal limit. But why were three of the pools totally fine? Just four of the seven had these crazy high levels of combined chlorine.
Related: How to reduce combined chlorine
In our site visit that lasted about three hours, we tried our best use forensics and evidence to solve the mystery. Based on what we could tell, there was virtually no difference in bather load between any of the seven pools, because bathers transferred from pool to pool constantly. One was a big wave pool, another was a splash pad, one was a giant set of slides into a catch basin, and a few others. So we went into the pump room to look at any possible place ammonia could be introduced to the water. We checked every chemical, and looked especially for ammonia-based algaecides, which are common in outdoor pools.
Nothing.
We double checked the water chemistry and sure enough, the combined chlorine was a major problem using both test kits. Finally, we remembered the competition pool from years before and asked to see the deck cleaning product used. The facility operator took us outside to the chemical storage room to find a 55-gallon drum of ZEP DZ-7 disinfectant floor cleaner, shown in the photo above. The active ingredients were all ammonia compounds. While the product is great at cleaning floors, the ammonia aspect of it makes it problematic for pool decks. So with that in mind, we went back into the waterpark.

Sure enough, we noticed the three pools without combined chlorine issues all had rope fences around them. These fences, shown in the photo above, prevented the floor scrubbing zamboni from getting too close to the water. Whereas the affected four pools had no such fence, and the zamboni was able to drive right over the gutters, which meant the ammonia deck cleaner was put directly into circulation every night!
To ZEP's credit, they (or their distributor) swapped out the product for a non-ammonia cleaning product for the waterpark, free of charge. Within a week, the combined chlorine had normalized and the problem was solved. And it's a good thing the combined chlorine levels were resolved, because the health department was going to shut down the entire facility. It was an urgent problem for the waterpark.
Conclusion
Any time a pool struggles with combined chlorine, the nitrogen/ammonia came from somewhere. To solve the issue, you must first find the source. Normal levels, like that of bathers alone, is usually able to be addressed by normal pool chemistry and secondary systems like UV, ozone, AOP or HDO. The combined chlorine levels that normal swimming pool chemistry and equipment cannot handle are usually caused by external sources. Most common among them are tap water chloramines and ammonia-based deck cleaners, such as dimethyl ammonium chloride.
Do what you can to address the source of the nitrogen/ammonia, and maintaining low levels of combined chlorine will be much easier. Since bathers introduce urea through sweat and urine, some combined chlorine is inevitable, but should be manageable. Tap water chloramines can be filtered out with catalytic carbon filters on the fill line. Ammonia deck cleaners, on the other hand, overwhelm pool chemistry, and should simply be replaced with a non-ammonia cleaning product instead.
1 Lowry, Robert W. (2009). IPSSA Intermediate Training Manual. Part 1-Chemicals. Page 57.
2 Beech, J.A. (1981). Chemistry in Water Reuse. Vol. 1, Chapter 20, "Pollution in Swimming Pool Water." Ann Arbor Science.
3 Lowry, Robert W. (2009). IPSSA Intermediate Training Manual. Part 1-Chemicals. Pages 64-65.

 By
By